Webinar: Chemistry & Hazardous Materials Management in SAP
Simple, flexible and compliant
WITH PRACTICAL EXAMPLE OF A GERMAN CHEMICAL COMPANY
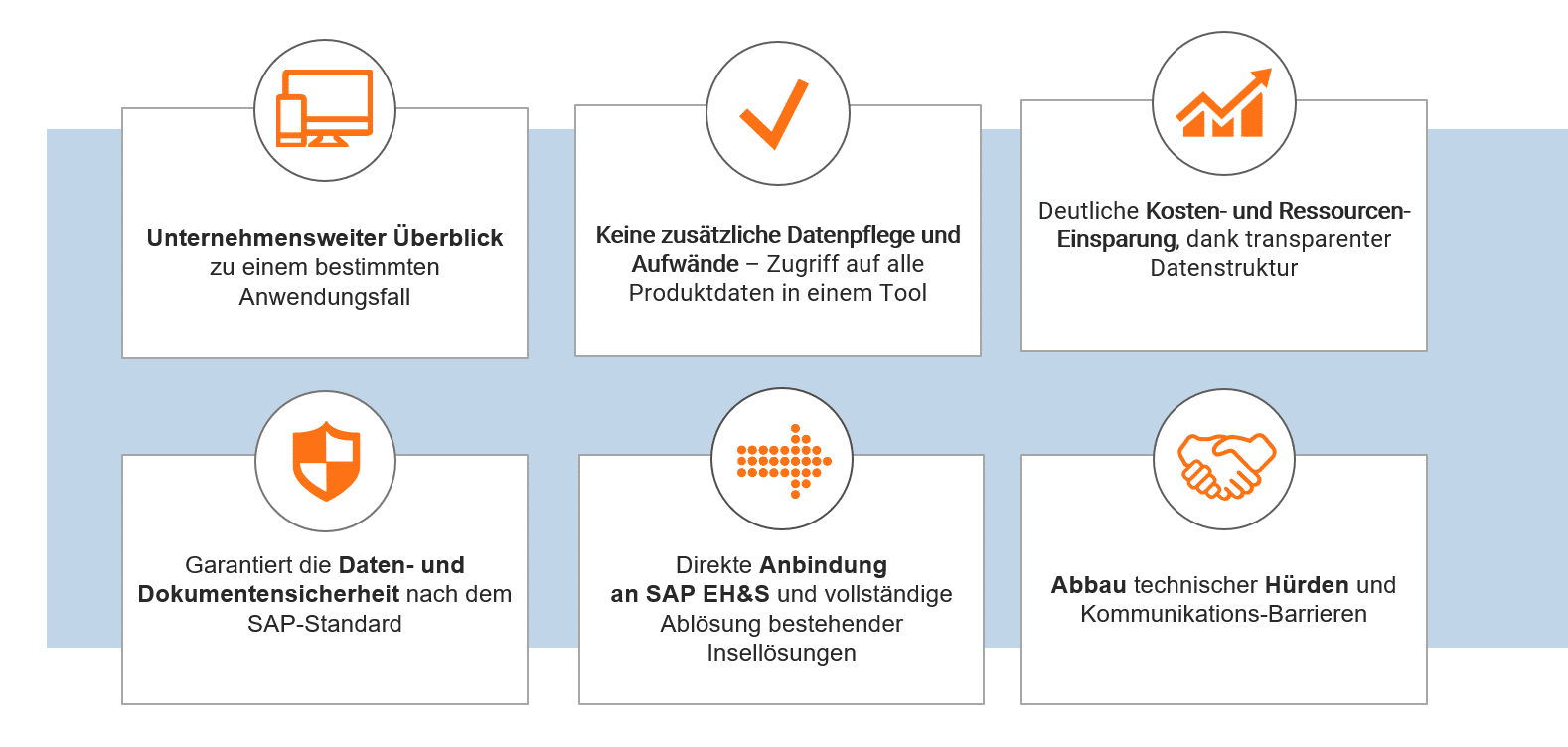
In the webinar, you will learn how quickly and easily products, studies or specifications from a wide variety of data sources and responsibilities can be easily merged in an SAP system - with modern and intuitive business apps. And all this while taking regulatory matters and product conformity into account.